Both manure and compost can improve soil tilth, but the timing and mechanisms may differ.
Cover crops and ridge tillage in sustainable farming.
In practice, cover crops such as legumes (e.g., clover, vetch) and grasses are often grown either to supply N to subsequent cash crops or to reduce nitrate leaching and soil erosion. Crop rotation and intercropping have been used to achieve this goal. Work in tropical humid regions of India has shown that corn yields and N uptake increased by 15 to 20% by intercropping with soybean or cowpea. Legume intercropping was equivalent to adding 80 kg N/ha of fertilizer N. A marked synchrony between plant growth stage and microbial population in the rhizosphere 60 days after seeding was observed in this study.
The N release from cover crops, however, depends on many factors, including the C to N ratio of the residue, its degree of incorporation into the soil and soil temperature and moisture regimes. It may take one to three weeks after cover crop incorporation before N release exceeds N immobilization. The farmer may have to decide if additional fertilizer N is needed and, if so how much. On the other hand, when the soil is not cropped, cover crops, especially grass, can reduce the potential loss of N through leaching or denitrification. In the US mainland, rye is often used as a winter cover crop to control weeds through competition and allelopathy.
Along with crop rotation and intercropping, ridge tillage is often practiced in sustainable farming. This technique only tills a zone of soil 15 to 20 cm wide and 5 to 10 cm deep at planting time. The practice creates a desirable seedbed environment, it also covers crop residue and weeds in the area between rows with soil. Subsequent between-row cultivation a couple of weeks after planting promotes mineralization of soil organic residues, controls weeds and reforms the ridge with a raised bed for future crops.
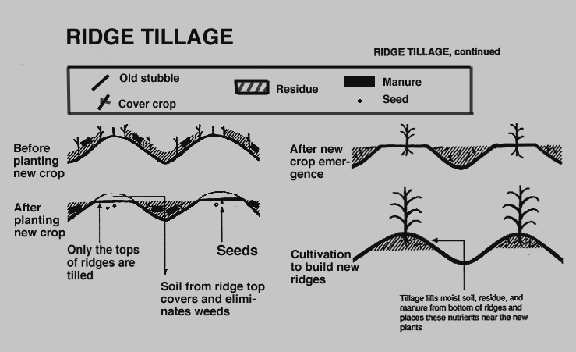
(Adapted from WSARE, 1995)
PHOSPHORUS.
Along with nitrogen, phosphorus is a nutrient that plants need in relatively large quantities for normal growth. In fact, P is a structural component of DNA and RNA, the two genetic entities that are essential for growth and reproduction of living organisms. Living organisms, whether plants or humans, also derive their internal energy from P-containing compounds, mainly adenosine diphosphate (ADP) and adenosine triphosphate (ATP). This means that inadequate P supply will result in a decreased synthesis of RNA, the protein maker, leading to depressed growth.
Phosphorus-deficient plants, therefore, are stunted with a limited root system and thin stems. In many plants such as corn, guava, and tomato, seedlings look stunted, and older leaves may turn purple because of the accumulation of anthocyanins or purple pigments. Fruit trees deficient in P have few and short new shoots and malformed fruits and seeds. Thus not only low yields but also poor quality are the results of P deficiency.


Internally, most crops need from 0.2% to 0.5% P in the dry matter for normal growth. Macadamia is an exception, iron chlorosis may occur if P content is greater than 0.15% in leaves.Table 2 lists the sufficiency range pf P in leaves of selected crops (Tamimi et al., 1994).
Table 2. Sufficiency P levels in leaves of selected crops.
| Crop |
Plant P, % |
| Cabbage |
0.4 - 0.6 |
| Coffee |
0.2 - 0.3 |
| Corn |
0.3 - 0.5 |
| Head lettuce |
0.4 - 0.6 |
| Macadamia |
0.08 - 0.10 |
| Soybean |
0.3 - 0.5 |
| Surgarcane |
0.2 - 0.3 |
| Tomato |
0.3 - 0.8 |
Unlike animals and humans who can obtain P from various food sources, plants must extract P exclusively from the soil where they grow. Also unlike humans whose gastrointestinal enzymes can break up organic P in food, plant roots can absorb P only when P is in the soil solution and in either H2PO4 - or HPO42- form. This fact is emphasized because not only do plants like H2PO4 - and HPO42- but so do soils. There is a strong competition between plants and soils for P in the soil solution. And the winner usually is soils, especially highly weathered soils like those of Hawaii and the tropics.Most of our soils here contain large amounts of iron- and aluminum-oxides or amorphous alumino silicate clays, which tie up P firmly, making P virtually unavailable for plant uptake. For example, in order to provide enough P for many of our favorite vegetables, such as lettuce and tomato, first time growers must apply approximately:
Table 3. P fertilization for good growth of most vegetables
| Soil |
P fertilizer |
| (Series / Order) |
(lb / A) |
| Hilo / Andisol |
2,000 |
| Halii / Oxisol |
1,600 |
| Paaloa / Ultisol |
1,200 |
| Lualualei / Vertisol |
100 |
(Adapted from Hue, 1991; and Hue et al., 1994)
Of course, amounts of P required would vary, depending on how much P the soil has, to begin with. That¹ s true, and that¹ s why you need a soil test before trying to amend or fertilize your soils. In fact, by adding P to the soil year after year, you might build up soil P to a point that it becomes detrimental to your crops because of excess. That was the case for several vegetable growers in the Waianai area of Oahu, Hawaii. The upside of this phenomenon is that once you have built your soil P to a good level, that level would remain for many years without any additional P input. The reason is that unlike nitrogen, P does not move easily with water. It virtually stays put. Leaching is minimal.
The next question is what can we use to build up soil P? In conventional farming you can use such fertilizers as treble superphosphate (Ca(H2PO4)2) which contains about 20% P or 46% as P2O5 or diammonium
phosphate (DAP) which has coincidently the same amount of P as in treble superphosphate. These fertilizers are highly soluble, dissolve quickly in water and can provide enough P to your crops before being tied up by the soil. These P fertilizers, however, are not acceptable in organic farming. Here we must rely on different sources.
Table 4. Total P concentration in organically acceptable sources
as compared with treble superphosphate (20% P).
| Source |
Total P, % |
| Rock phosphate |
17 - 26 |
| Bone meal |
20 - 30 |
| Fish meal |
5 - 10 |
| Wood ash |
2 - 5 |
| Poultry manure |
0.5 - 1.5 |
| Green maunure |
0.2 - 0.5 |
| Compost |
0.2 - 0.5 |
| Sewage sludge |
0.4 - 2.5 |
(Adapted from Nick and Bradley, 1994; and Hue, 1995)
Table 4 shows P content of selected sources that are acceptable to organic farming community in most states. Among these, the first two, rock phosphate and bone meal, have reasonably high total P content: between 20 and 30%. However, P in these two sources is very insoluble, thus much less plant available than P in treble superphosphate. More specifically, P in rock phosphate and bone meal has the formula: Ca5(OH)(PO4)3 which is hydroxy apatite or apatite for short. This is the same material that our bones and teeth are made of. As our teeth can attest to it, apatite is quite durable; is very hard to dissolve in water, meaning that it provides very little phosphate to your crop in the short term.
A couple of things should be mentioned about rock phosphate. First, it is a source of P for long-term soil improvement, don¹ t expect any noticeable effect from it within weeks or months, unless you use huge amounts of it. Second, its solubility, and thus plant availability, depends strongly on soil pH and particle size. It is more effective in acid soils than in calcareous or alkaline soils; more effective when it is fine than coarse, and more effective in the presence of mycorrhiza than without mycorrhiza.
By contrast, P contents of chicken manure, compost, and sludge are relatively low, usually below 3%. Thus, large amounts would be needed to meet P requirement of the crop. Yet, pound for pound, P from these organic sources is quite available to plants; sometimes even more effective than treble superphosphate. Our recent work has shown that organic matter from the manure interacts with clay minerals and reduces P sorption by the soil, thereby enhancing P availability to plants (Hue, 1990; 1991).
POTASSIUM.
Compared favorably with nitrogen, potassium is needed in large quantities by many crops. Table 5 lists the sufficiency range of K in selected crops.
Table 5. Sufficiency range of K in selected crops.
| Crop |
Tissue K, % |
| Banana |
3.0 - 5.0 |
| Cabbage |
4.5 - 7.5 |
| Coffee |
2.0 - 3.0 |
| Corn |
1.5 - 3.0 |
| Lettuce |
4.0 - 7.5 |
| Papaya |
3.0 - 5.0 |
| Tomato |
3.0 - 5.0 |
(Adapted from Tamimi et al., 1994)
In plants, potassium is required for maintaining osmotic potential of cell. That is, K makes plants look turgid. Since K regulates the osmotic potential of cells, and the close or open conditions of stomata, it plays an important role in water relations in the plant. Potassium is involved in water uptake from the soil, water retention in the plant tissue, and long distance transport of water in the xylem and of photosynthates in the phloem. Potassium affects
cell extension. With adequate K, cell walls are thicker, thereby improving plant resistance to lodging, pests and disease. Fruits and vegetables grown with adequate K seem to have a longer shelf life in the grocery store.
Consequently, K-deficient plants show low resistance to disease, their seeds and fruits are small and shriveled. In tomato, K deficiency results in smaller fruits whose flesh development is incomplete. In corn, the maturity is delayed and ears are smaller when K is deficient.


(Source: Potash and Phosphate Institute, Atlanta, GA)
The stalks are weak and lodging is common. The most visual K deficiency symptom is the scorching or firing along leaf tips and margins as shown here for soybeans. In alfalfa and clovers, the first sign of K deficiency shows up as small white or yellowish dots around the outer edges of leaves. As the deficiency intensifies, these edges turn yellow, then brown, and finally die.


(Source: Potash and Phosphate Institute, Atlanta, GA)
In soils, potassium is quite mobile as compared to phosphate. It exists as K+ in soil solution and is absorbed by roots in that form. Although K+ can be retained to some extent by negative charges on clay surface, it can be displaced into the soil solution by Ca2+ or Mg2+, when gypsum or dolomite is added. Thus if K is not taken up by plants, it might be lost by leaching. One way to reduce K leaching is to add organic matter such as compost or green manure to your soil. These organic materials usually have large cation exchange capacity which can retain K effectively. Among soils, K deficiency occurs more often in sandy than in clayey soils, more often in highly weathered soils than in young soils, and more often in volcanic ash-derived soils of the Hilo coast than those of the Kona coast.
If your soil is low in K, what can you do? Commercial KCl is an easy choice in conventional farming, but not acceptable in organic farming. What alternatives do we have?
Table 6. Total K concentration in selected organically acceptable sources.
| Source |
Total K, % |
| Sul-Po-Mag [Mg, K, SO4] |
22.0 |
| Polyhalite [Ca, K, SO4] |
10 - 15 |
| Wood ash |
5 - 10 |
| Green sand |
5 - 7 |
| Green manure |
2 - 5 |
| Seaweed meal |
2 - 3 |
| Compost |
0.5 - 2.0 |
(Adapted from Nick and Bradley, 1994; and personal data)
The first two sources are natural deposits of potassium minerals. Sul-Po-Mag as the name implies is a double salt of magnesium and potassium sulfates. It has about 22% K. Polyhalite is a double salt of Ca and K sulfates. It has about 10 to 15% K. Like Sul-Po-Mag , polyhalite is very soluble, not much different from KCl. In terms of plant response, this material is as good as or even better than KCl or gypsum (unpublished data). There are large deposits of polyhalite in Utah, Texas, Poland, and Egypt. How commercially available is polyhalite to sustainable farmers? I do not really know.
Wood ash also contains considerable amount of potassium, and this source is also readily available (and alkaline). On the other hand, green sand has about 7% K but the availability of this K is very low. It can be used to build up K reserve for the soil, but I doubt very much that it can meet the K requirement of your fast growing crop.
It is hoped that the information provided would be useful to you in managing your soil or in assisting your clients. One thing I would like to repeat is that sustainable agriculture is more in
tune with nature than conventional agriculture, but it is information intensive and requires greater management skills from the farmers than ever before.
Table 7. General properties of some organic fertlizers
References
- Bugg, R. L. and P. R. Miller. 1991. Grasses/legumes:Dream team. Sustainable Agric. News 3(4):1-4. UC Davis, CA.
- Caplan, B. 1992. Organic gardening. Headline Book Publ., London, UK.
- Hue, N. V. 1990. Interaction of Ca(H2PO4)2 applied to an Oxisol and previous sludge amendment:soil and crop response. Commun. Soil Sci. Plant Anal. 21:61-73.
- Hue, N. V. 1991. Effects of organic acids/anions on P sorption and phytoavailability in soils with different mineralogies. Soil Sci. 152:463-471.
- Hue, N. V., H. Ikawa, and X. Huang. 1994. Predicting phosphorus requirements of some Hawaii soils. Fact Sheet no. 2. CTAHR.
- Hue, N. V. 1995. Sewage sludge. pp. 193-239. In J. E. Rechcigl (ed) Soil amendment and environmental quality. Lewis Publ., Boca Raton, FL.
- Nick, J. and F. Bradley. 1994. Growing fruits and vegetables organically. Rodale Press, Emmaus, PA.
- Tamimi, Y., J. A. Silva, R. S. Yost, and N. V. Hue. 1994. Adequate nutrient levels in soils and plants in Hawaii. Fact Sheet no. 3. CTAHR.
- Today's Chemist. 1995. May issue. Am. Chem. Soc.
- WSARE. 1995. The basic principles of sustainable agriculture. URL: http://ext.usu.edu/wsare (the internet).